As the world battles the Wuhan coronavirus pandemic, ventilators are in high demand and short supply. Existing manufacturers have ramped up production of the devices, which utilize proprietary technology, but absent a disruptive event a major shortfall is still projected.
At least one disruptive event is taking place, enabled by a loosening of government regulations and rewarding innovation. (Shocking, right?)
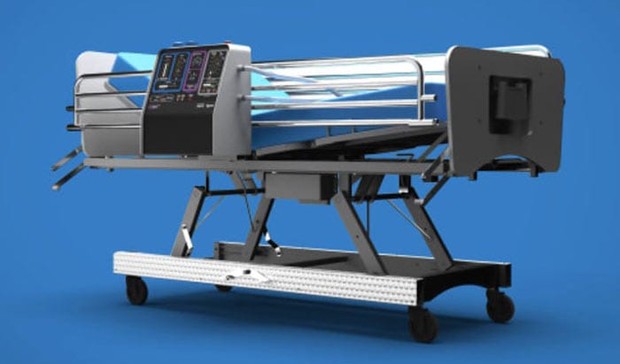
U.K.-based Dyson, known for its vacuum cleaners, hair dryers, and air purifiers, has designed an entirely new ventilator system in just 10 days and has agreed to produce 15,000 of them. The ventilator, named CoVent, (did you see what they did there?) is “a bed-mounted, portable ventilator that can run from battery power in field-hospital conditions.”
Working under a grant from the U.K. government, with oversight from the U.K. National Health Service and the Medicines and Healthcare products Regulatory Agency (MHRA), Dyson has agreed to produce 10,000 ventilators for the country. On top of that, it will produce another 5,000 ventilators for donation. One thousand of those will go to the U.K. The remaining 4,000 will go to other countries. CoVent will need to receive regulatory approval before receiving funding and going into production.
In an email to employees James Dyson, the company’s founder, said that “the new device can be manufactured quickly, efficiently and at volume.” A company spokesperson told CNN that, pending speedy regulatory approval, “the ventilators would be ready by early April.”
Here is the complete text of the email Dyson sent to employees Wednesday (from Fast Company):
Hospitals are the frontline in the war against Covid-19, where heroic doctors, nurses, and care workers are battling to save lives and help people recover from this terrible virus. As with any battle, there are many challenges to overcome, not least the availability of essential equipment which in this case means ventilators. A ventilator supports a patient who is no longer able to maintain their own airways but sadly there is currently a significant shortage, both in the UK and other countries around the world.
Since I received a call from Boris Johnson ten days ago, we have refocused resources at Dyson, and worked with TTP, The Technology Partnership, to design and build an entirely new ventilator, The CoVent. This new device can be manufactured quickly, efficiently and at volume. It is designed to address the specific clinical needs of Covid-19 patients, and it is suited to a variety of clinical settings. The core challenge was how to design and deliver a new, sophisticated medical product in volume and in an extremely short space of time. The race is now on to get it into production.
The Dyson Digital motor sits at the heart of the new device and the motor’s design is optimised to have a very high level of intrinsic safety, making it particularly well-suited for industrial, high volume production. The device is designed to achieve a high quality air supply to ensure its safety and effectiveness, drawing on our air purifier expertise which delivers high-quality filtration in high-volume products.
Ventilators are a regulated product so Dyson and TTP will be working with the MHRA and the Government to ensure that the product and the manufacturing process is approved. We have received an initial order of 10,000 units from the UK Government which we will supply on an open-book basis. We are also looking at ways of making it available internationally.
I am proud of what Dyson engineers and our partners at TTP have achieved. I am eager to see this new device in production and in hospitals as soon as possible. This is clearly a time of grave international crisis, I will therefore donate 5,000 units to the international effort, 1,000 of which will go to the United Kingdom.
We will keep you updated with our progress.
Best wishes,
James
In the United States, President Trump tweeted that auto makers “Ford, GM, and Tesla are being given the go-ahead to make ventilators and other medical products,” as the FDA published a new guidance that “not only gives medical professionals broader latitude to modify existing FDA-approved ventilators, it also creates a streamlined process for complete newcomers to the ventilator market to get FDA approval.”













Join the conversation as a VIP Member