Medical research on human subjects it is an area of science that is fraught with ethical peril. Patients must be fully informed of all benefits and risks through a process of informed consent. But all human subjects are not alike. An educated person suffering from a relatively mild condition can probably make a solid choice for their own treatment. A person with a life threatening disease will often grasp at straws. Witness the Laetrile clinics that once populated the US-Mexico border and preyed upon desperate people. Even smart and well connected people put their commonsense in a lockbox in life and death situations. Steve Jobs, for instance, probably shortened his own life by relying on “alternative” medicine. When a parent is trying to save a extremely ill and fragile child the same effect kicks in. When you couple this with the way were are culturally taught to trust doctors and nurses, you have a population that must be treated as extremely vulnerable to abuse.
When desperate people are confronted with doctors working with the imprimatur of the US government, the ability to rationally evaluate risks and benefits is further eroded. When the risks to the patients are explained in a blizzard of forms written with a view to obfuscate rather than enlighten then the patient has no plausible way of making an informed decision.
Like so many really bad stories, this one begins with the bests of intentions. And because all the intentions are good, nothing bad can happen, right?
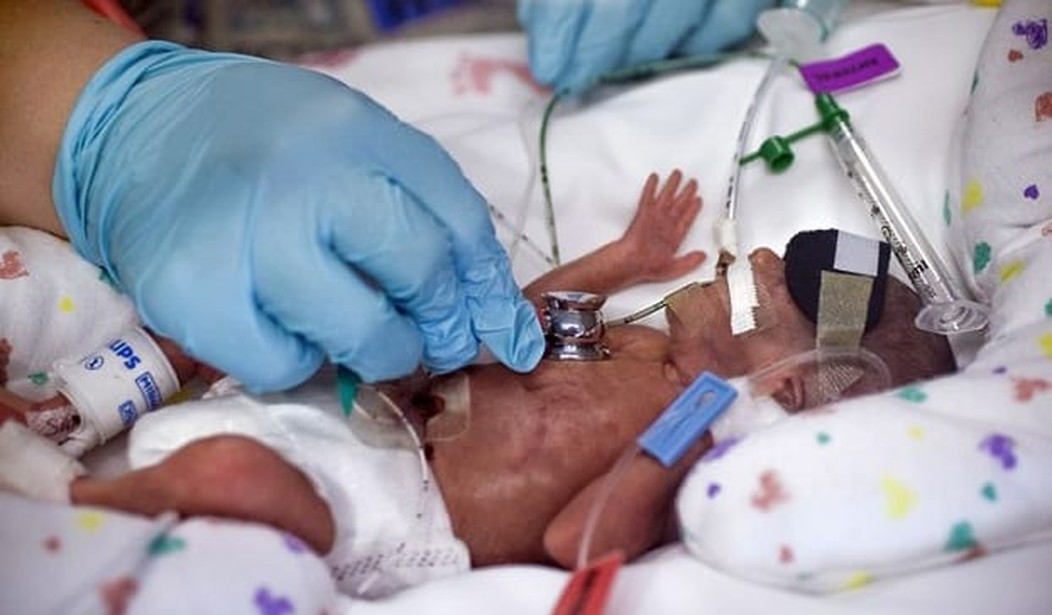
Premature infants are, needless to say, very delicate. They are often born before organs completely develop and the treatment they receive during those initial weeks of life determine, to a great extent, the quality of life they will have as they mature. Not only are they delicate, typically parents are distraught. One of the areas most problematic and least understood is the application of supplemental oxygen. Oxygen is given to virtually all premature babies. Too much results in eye damage or blindness. Too little, death.
In 2005, the National Institutes of Health (specifically the Eunice Kennedy Shriver National Institute of Child Health and Human Development) began a study called Surfactant Positive Airway Pressure and Pulse Oximetry Trial (SUPPORT) in an effort to determine how much supplemental oxygen to administer.
To do so they recruited the parents of 1316 extremely premature infants who were born between 24 weeks 0 days and 27 weeks 6 days of gestation. The idea was to find out the ideal range of supplemental oxygen for premature babies. The gold standard for science, at least outside of the modern phrenology called “climatology,” is the controlled double blind study. To achieve that goal it appears that the researchers deviated from treatment norms:
The NIH-funded experiment used the test babies in an attempt to find the sweet spot for preemies yet to be born: the lowest level of oxygen that would preserve vision, yet be sufficient to prevent brain damage and death.
To get the answer, researchers arbitrarily assigned infants to either a high-oxygen or low-oxygen group. Because, researchers say, all oxygen levels fell within the generally accepted range, they argue the babies received the same “standard of care” as babies not in the study. None of the consent forms mentioned a risk of death from the oxygen experiment.
But it turns out there were key differences in how researchers treated babies in the study compared with those not in the study.
Normally, medical personnel constantly adjust oxygen as preemies’ conditions change, based on their individual needs. But the SUPPORT study was designed to keep infants in their randomly assigned range, despite a baby’s individual needs.
And in a decision that one government source says shocked seasoned researchers when they learned of it, the babies’ oxygen monitors intentionally were altered to provide false readings. The reason: so medical staff wouldn’t be tempted to adjust oxygen out of the babies’ study-assigned range.
The results have left participating parents with a lot of questions:
It was Oct. 11, 2006. Medical personnel asked Cook, then a 26-year-old single mother, to enroll little Dreshan in a study. She says they described it as a program offering assistance and encouragement to preemies—premature babies—and their families. She readily signed the consent form.
“I remember them telling me they were a support group who would pretty much hold my hand through the developmental process,” Cook says.
…
All three women now say they never would have agreed to take part if they had known the NIH-funded study’s true nature—to randomly manipulate preemie oxygen levels. They discovered that just last year.
Dreshan and Christian are now 7 years old and both struggle with myriad health problems. Destiny died within three weeks. The mothers wonder: Did the experiment contribute to any of the medical problems of their children?
“[Dreshan] was already at a slim chance of surviving; why would I make his chances of surviving more slim?” Cook asks.
The NIH has made a defense, though a lame and legalistic one. In essence they say that 1) the consent form was properly approved and 2) all the infants received supplemental oxygen in the normal range. What they pass over is that the Office of Human Research Protections (OHRP), the people charged with ensuring all human subjects research comports with the law, said the disclosure was misleading and that in order to achieve the correct environment for experimentation, the past practice of adjusting oxygen level based on the individual child was suspended.
Public Citizen is now calling for an investigation into the structure of the study and interference by senior NIH officials in the process of protecting human subjects:
Public Citizen, joined by nine prominent scholars, today called for an immediate investigation by the U.S. Department of Health and Human Services (HHS) Office of Inspector General into the conduct of senior HHS officials, who, according to an email trail, permitted top National Institutes of Health (NIH) officials to edit drafts of a letter documenting findings of what should have been an independent inquiry into serious ethical lapses in a major NIH trial.
According to documents Public Citizen recently obtained under the Freedom of Information Act (FOIA), HHS officials in the immediate Office of the Secretary and Office of the Assistant Secretary for Health (OASH) knowingly allowed the director of NIH and other senior NIH officials to interfere with the independence of the Office for Human Research Protections’ (OHRP’s) ongoing compliance oversight investigation of the controversial SUPPORT study, Public Citizen said in a letter to the HHS inspector general.
In a separate letter to the HHS inspector general, U.S. Rep. Rosa DeLauro (D-Conn.) today echoed the call for an investigation.
Though heavily redacted, the documents Public Citizen obtained reveal that named NIH officials were, inappropriately, given multiple opportunities to review and edit drafts of a pending OHRP compliance oversight determination letter regarding the SUPPORT study, as well as apparently allowing NIH to influence the timing of the release of the letter, which occurred on June 4, 2013. This letter put on hold all compliance enforcement actions taken by OHRP that had been outlined in an earlier letter issued on Feb. 8, 2013, to the University of Alabama at Birmingham. This hold is still in effect.
This story points to a distressing trend in American medical research. Large amounts of money and many careers are invested in high profile studies. Just as the NIH bullied the OHRP into changing its critical report, so, too, are university Institution Review Boards, the bodies responsible for protecting human and animal subjects at research universities, are also susceptible to political pressure to massage the rules in order to allow research. As we enter an era where our medical community is increasingly non-Chrisitan and irreligious (as an aside here, when Francis Collins was nominated to head NIH there was widespread protest in the research community because Collins is an Evangelical Christian, for example here), pursuit of scientific knowledge, unencumbered by ethical restraints, becomes a siren song that is difficult to resist as we’ve seen in the saga of embryonic stem cell research.
If true, this is troubling. Absent truly informed consent we are on a glideslope to return to the days of the Tuskegee experiment. If the agency funding problematic research is given carte blanche to edit oversight reports then there are no longer any safeguards available for patients participating in medical research.
For more on this read Sharyl Atkisson’s series at The Daily Signal.












Join the conversation as a VIP Member